[Commonwealth Magazine published this analysis and commentary on May 4 2019.]
Many Bay State health care cognoscenti and politicos like to brag about Massachusetts health statistics. For years now, Massachusetts has performed well, at or near the top, in surveys of key health indicators among the 50 US states.
For example, the United Health Foundation’s 2016 America’s Health Rankings had Massachusetts in 1st place (though we dropped to 7th in 2018). We were 2nd in the Commonwealth Fund’s State Health System Performance Scorecards in 2018. And we showed up 5th in the U.S. News & World Report’s Best States survey. Not too shabby.
Maybe we should limit the self-congratulations. Perhaps we’re not as good as we like to believe. What if comparing ourselves with retrograde US states sets the bar too low? By contrast, the Massachusetts education policy community routinely examines benchmarks comparing our state’s performance with that of other advanced nations, not with US states where looking smart is no big challenge. Here’s a recent example:
“If Massachusetts were a nation, it would share the top spot in reading with eight other nations worldwide. In science, the state’s students and those from 10 nations came in second, trailing only students from Singapore. In math, 11 other nations were ahead of the Commonwealth. The results come from the 2015 Program for International Student Assessment (PISA), a triennial international survey designed to assess how well 15-year-old students can apply their knowledge and skills.”
So, how does Massachusetts compare on key health statistics with those of other advanced nations? Are we tops? Do we win the crown or not?
Not.
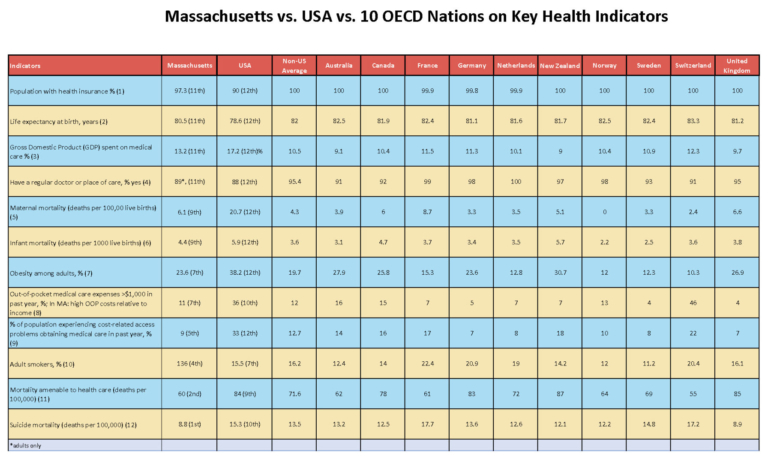
With research assistance from a diligent graduate student, I examined 12 key health performance indicators for Massachusetts and matched them with comparable stats from 11 advanced nations: the US, Australia, Canada, France, Germany, the Netherlands, New Zealand, Norway, Sweden, Switzerland, and the United Kingdom. I included core public health measures often included in international and US comparative performance studies:
Obesity among adults
Adult smoking
Population with health insurance
Infant mortality
Life expectancy at birth
Share of gross domestic product (GDP) spent on medical care
Maternal mortality
Suicide mortality
Having a regular physician or place of care
Mortality attributable to health care
Population experiencing cost-related access problems
Population with out-of-pocket health care costs greater than $1,000 in past year
Some argue that it is illegitimate to compare a nation as large as the US with comparatively puny competitors. For comparative purposes, the US population in 2017 was 325.7 million, and the 10 non-US comparators’ combined population was 322.8 million. For this analysis, I examined the 10 non-US nations as a group and individually with the US and with Massachusetts – 12 categories in all. The accompanying table provides data and rankings for Massachusetts, the US, and the average of the other 10 nations. (To see the full table with sources and with details on all 11 examined nations and Massachusetts, click here.)
How does the US come out? On the 12 measures among 12 nations (treating Massachusetts as a nation), the US ranks 12th worst on 8 measures, 10th worst for 2 measures, and 9th and 7th worst for 1 measure each. Looking at the three units – 10 nations, US, and Massachusetts – our nation comes in last on 11 of 12 measures, and best on zero.
What about Massachusetts? On 8 of the 12 measures, we’re in the bottom half; on 4 of those, we come in at #11, one rank better than the US, and worse than everyone else. We’re 11th best out of 12 on health insurance coverage, life expectancy, share of Gross Domestic Product spent on medical care, and having a regular physician or place of care. We are 9th best on maternal mortality and infant mortality.
On the other hand, we are best among the 12 on having a low suicide rate, and 2nd best on mortality attributable to medical care. On the rest, we are in the middle of the pack. When just looking at the 10 non-US nations collectively, the US, and Massachusetts, we are best on 5 indicators, and worse than our competitor nations on 7, though better on all of these than the US.
Surprises? I incorrectly expected that Massachusetts would be better than 4th on adult smoking. I did not realize that the Massachusetts suicide rate would be so positive. It is remarkable that while Massachusetts has the highest rate of health insurance coverage among all 50 states, at 97.3 percent, our rate is lower than the rates in all 10 non-US nations.
Because Massachusetts has such a high level of spending on medical care, I expected we would spend a larger proportion of our state’s GDP on health care than the US and come in dead last. Instead, we’re 11th. What explains this? It’s not so much the numerator (health care spending), as it is the denominator (the state’s high total GDP) which reflects a far more affluent state than most of the other 49. Even though our spending looks high, it is lower than the US average in its burden.
Looking to education policy as a model, Massachusetts should be less concerned with comparisons to other states’ performance, and more attuned to comparing our results with those of other advanced nations. Massachusetts policy experts would do well to pay closer attention to factors that influence the superior performance of these nations to ours. If other nations can kick our butts so convincingly on maternal and infant mortality, life expectancy, health care spending, and other essential measures, then we should focus more on how we can close the gap with these nations than comparing ourselves with our fellow states.
For the past decade, since passage of the state’s 2006 universal health care law and the 2012 cost containment law, Massachusetts has focused on controlling health care cost increases. While this has been a valuable and successful effort, I believe it also has crowded out attention to key determinants of health, especially obesity, that drive up health care spending substantially and harm public health. Perhaps it is time for the Commonwealth to reassess its core health system priorities.




